Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Takano, Masahide; Ito, Akinori; Akabori, Mitsuo; Minato, Kazuo; Numata, Masami
Proceedings of GLOBAL2003 Atoms for Prosperity; Updating Eisenhower's Global Vision for Nuclear Energy (CD-ROM), p.2285 - 2291, 2003/00
Stability of AmN and (Am,Zr)N was studied comparatively from the viewpoints of the hydrolytic and evaporative behavior. AmN powder reacted with moisture to form hydroxide Am(OH) , while the solid solution (Am
, while the solid solution (Am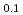 Zr
Zr )N remained stable as long as 1000 hours. Stabilization effect of ZrN was found to depend significantly on its mole fraction from the experiments on (Dy,Zr)N. In the oxidation experiments on (Dy,Zr)N by TG-DTA technique, rapid weight gain by the oxidation occurred above 700 K. Effect of ZrN on the stability against oxygen was slight. Nitrogen release by the evaporation of AmN and (Am
)N remained stable as long as 1000 hours. Stabilization effect of ZrN was found to depend significantly on its mole fraction from the experiments on (Dy,Zr)N. In the oxidation experiments on (Dy,Zr)N by TG-DTA technique, rapid weight gain by the oxidation occurred above 700 K. Effect of ZrN on the stability against oxygen was slight. Nitrogen release by the evaporation of AmN and (Am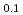 Zr
Zr )N in He gas flow was measured by gas chromatography. Evaporation rate constants of AmN were obtained at 1623-1733 K. Although the evaporation rate constant of AmN in the solid solution were lower than those of the pure AmN, the selective evaporation of AmN from the solid slution were recognized, which resulted in a decrease in the Am mole fraction.
)N in He gas flow was measured by gas chromatography. Evaporation rate constants of AmN were obtained at 1623-1733 K. Although the evaporation rate constant of AmN in the solid solution were lower than those of the pure AmN, the selective evaporation of AmN from the solid slution were recognized, which resulted in a decrease in the Am mole fraction.
Ito, Akinori; Akabori, Mitsuo; Takano, Masahide; Ogawa, Toru; Numata, Masami; Itonaga, Fumio
Journal of Nuclear Science and Technology, 39(Suppl.3), p.737 - 740, 2002/11
no abstracts in English
Ogawa, Toru; Omichi, Toshihiko; Maeda, Atsushi; Arai, Yasuo;
Journal of Alloys and Compounds, 224, p.55 - 59, 1995/00
Times Cited Count:23 Percentile:78.26(Chemistry, Physical)no abstracts in English